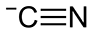SWEAT OF THE SUN



.jpg?crc=4001225444)
.jpg?crc=674159)




















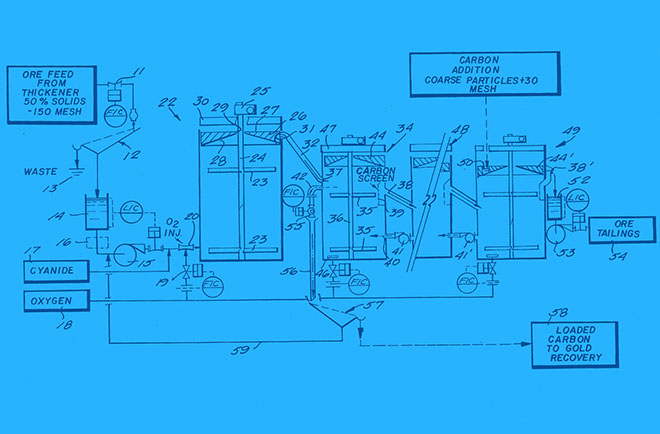
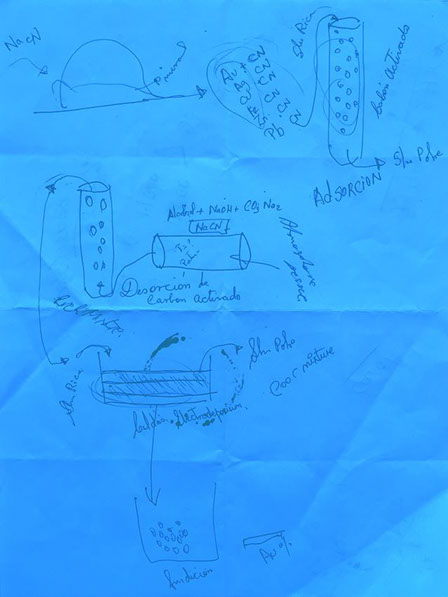
CARBON IN LEACH (CIL)
CIL is a method of gold extraction that uses charcoal as part of a standard cyanidation process. The process starts in SOTRAMI and ends at ‘Remega’ in Lima, a laboratory that removes both the cyanide and the gold from the charcoal.
SORTING
The gold ore is sorted on the surface by the Pallaqueras women and then weighed and crushed.
CYANIDATION
The crushed ore is dissolved through the method of ‘cyanidation’, where cyanide is used to transform the solid material into a liquid gold-cyanide solution, separating the gold from the ore.
ACTIVATION
The solution is mixed with ‘activated charcoal’ (processed charcoal to increase adsorption) in a ‘leach tank’, whereupon gold within the solution attaches itself to the carbon molecules (the gold and carbon have opposing ion charges).
ELECTROWINNING
The product is then treated with sulphuric acid before undergoing ‘electrowinning’ which utilises large-scale electroplates that attract the gold stripped by the caustic cyanide solution for retrieval. Within the electrolytic cell, an electric current passes from an inert anode through the liquid solution, attracting and depositing the gold onto the cathode in the process.
HEATING
The gold is then heated, melted and poured into moulds.
ELUTION
Impurities are removed from the loaded carbon under a cold and hot wash and then is transferred to the ‘elution phase’ or desorption of gold. This involves the use of hot caustic-cyanide (1%NaOH, 0.1 to 0.2%NaCN) solution that is kept at nearly boiling temperature for 36 to 72 hours. The elution rate is accelerated by adding alcohol and by pressure.













One of the myths surrounding the properties of gold is its ability to fluoresce. Throughout the history of gem mining, many lighting methods for prospecting minerals have relied on the notion of gems radiating light in the dark as the miner shines an ultraviolet lamp in their search. While a small number of minerals do indeed have a visible fluorescence under ultraviolet light, gold, however, is not one of them.